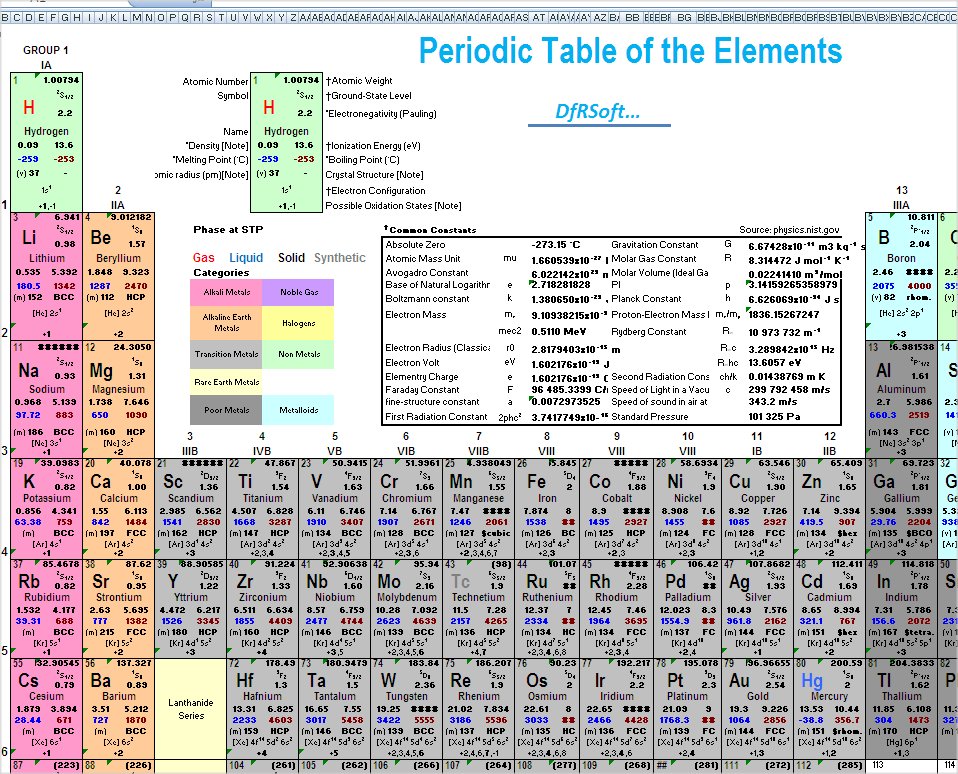
NGSS Claim Statement: Use the patterns associated with the location of elements in the Periodic Table to identify the number of portons and electrons present in several ions and relate the patterns to the tendency to form a stable octet of electrons.Įach atom has a charged substructure consisting of a nucleus, which is made of protons and neutrons, surrounded by electrons. Students earn the Trophy for this activity once they demonstrate mastery on all four Question Groups. The statements pertain to patterns associated with ion formation for main group elements. Once done, students must identify four statements as being true or false. First students must identify the number of protons and electrons in an ion of a main group element. This activity consists of 16 forced-choice questions organized into four Question Groups. Part 1: The Sub-Structure of Atoms and Ions The standards are the property of the Next Generation Science Standards. We are simply including them here for convenience (and because we have referenced the by number). Science and Engineering Practices || Crosscutting Concepts Disclaimer: The standards are not our original work. You can cross-reference the specific notations that we have used with the listings found on the following pages: The core ideas, crosscutting concepts, and science and engineering practices that we reference in our analysis are numbered for convenience. Here is our NGSS-based analysis of each individual activity of the The Periodic Table, Ions, and Ionic Compounds Science Reasoning task. Much of science deals with constructing explanations of how things change and how they remain stable. The functions and properties of natural and designed objects and systems can be inferred from their overall structure, the way their components are shaped and used, and the molecular substructures of its various materials. SEP 6.3: Constructing Explanations and Designing SolutionsĪpply scientific ideas, principles, and/or evidence to provide an explanation of phenomena and solve design problems, taking into account possible unanticipated effects.ĭifferent patterns may be observed at each of the scales at which a system is studied and can provide evidence for causality in explanations of phenomena. The repeating patterns of this table reflect patterns of outer electron states.ĭevelop, revise, and/or use a model based on evidence to illustrate and/or predict the relationships between systems or between components of a system.ĭevelop and/or use a model (including mathematical and computational) to generate data to support explanations, predict phenomena, analyze systems, and/or solve problems. The periodic table orders elements horizontally by the number of protons in the atom’s nucleus and places those with similar chemical properties in columns. Each atom has a charged substructure consisting of a nucleus, which is made of protons and neutrons, surrounded by electrons.The following DCI, SEPs, and CCCs are addressed at some point within The Periodic Table, Ions, and Ionic Compounds:ĭCI: PS1.A: Structure and Properties of Matter That is, the task of answering the question requires that the student utilize at least two of the three dimensions of the NGSS science standards - a DCI, a CCC, and/or an SEP. Each question is either a 2D or (preferrably) a 3D question. There are 53 multi-part questions organized into 22 Question Groups and spread across the five activities. Use the periodic table as a model to predict the relative properties of elements based on the patterns of electrons in the outermost energy level of atoms.Īs a whole, the questions in this task address a wide collection of disciplinary core idea (DCI), crosscutting concepts (CCC), and science and engineering practices (SEP). Collectively, the five parts were designed to address the following NGSS performance expectation: Each part involves a different type of skill or understanding. This NGSS-inspired task consists of five parts. Each activitiy places an emphasis upon the connection between the chemistry concept and the family properties of elemental groups on the Periodic Table. The activity addresses the HS-PS1-1 Performance Expectation of the Next Generation Science Standards. Finally, the last two activities pertain to the ratio by which atoms of elements combine to form ionic compounds. Then students explore the formation of ions by atoms of main group elements. The Periodic Table, Ions, and Ionic Compounds includes five scaffolded activities that begin with an investigation of the substructure of atoms and ions. Reasoning Center » Chemistry » The Periodic Table, Ions, and Ionic Compounds » About About The Periodic Table, Ions, and Ionic Compounds
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
